72+ pages depression in freezing point for 1 m urea 2.8mb. Apne doubts clear karein ab Whatsapp par bhi. 477 K views 24 K people like this Like Share. The depression in freezing point for 1M urea 1M glucose and 1 M NaCL are in the ratio Welcome to Doubtnut. Check also: point and understand more manual guide in depression in freezing point for 1 m urea K f 2.
Depression in freezing point for 1 M urea 1 M NaCl and 1 M CaCl_2 are in the ratio of. We have T f i K f m.

A 0 1m Urea Solution Shows Less Depression In Freezing Point Than 0 1m Mgcl2 Solution Why Quora
| Title: A 0 1m Urea Solution Shows Less Depression In Freezing Point Than 0 1m Mgcl2 Solution Why Quora |
| Format: ePub Book |
| Number of Pages: 143 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: November 2020 |
| File Size: 1.6mb |
| Read A 0 1m Urea Solution Shows Less Depression In Freezing Point Than 0 1m Mgcl2 Solution Why Quora |
 |
Depression in freezing point for 1 M urea 1 M NaCl and 1 M CaCl_2 are in the ratio of.

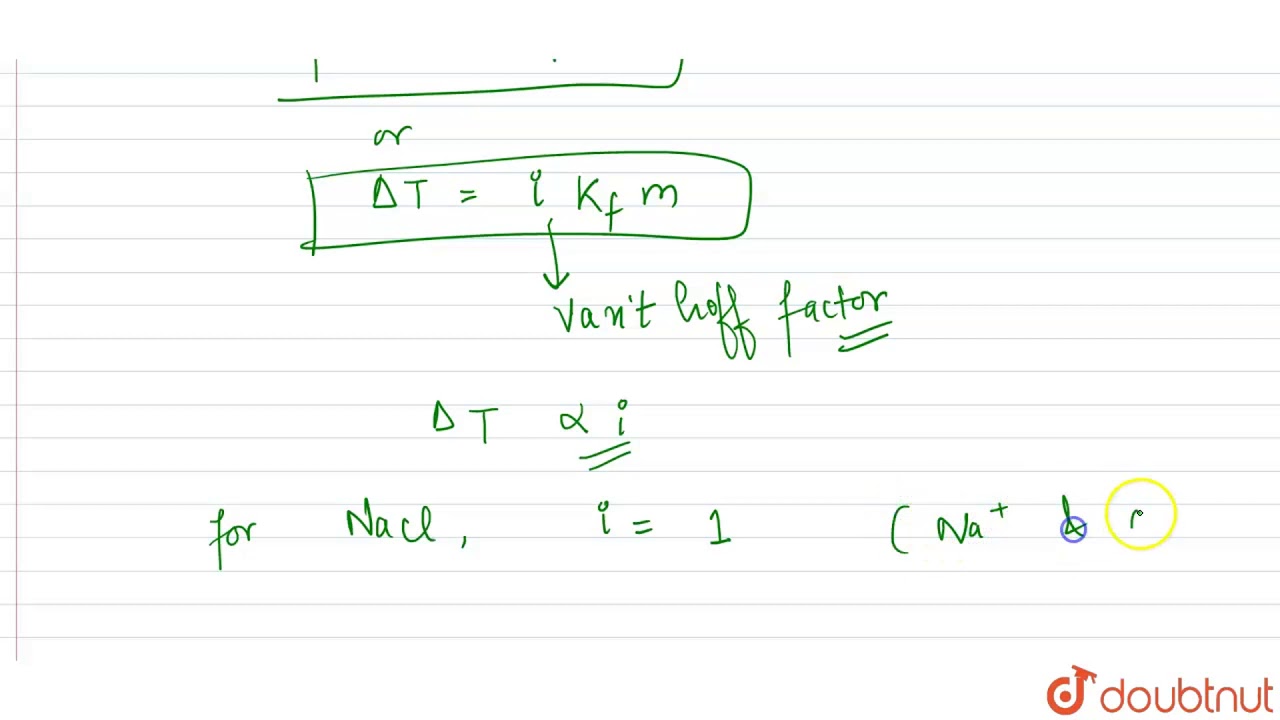
972 K views 684 K people like this Like Share. Depression in freezing point for 1 M urea 1 M NaCl and 1 M CaCl_2 are in the ratio of. As both 01 M solution of glucose and 01 M solution of urea contain same number of moles number of particles therefore both will have same depression in freezing point. The depressions in freezing point for 1 m urea 1 m glucose and 1 m NaCl are in the ratio. 0 m o l 1 k g and molaritymolality. I u r e a i g l u c o s e 1 i N a C l 2.

The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration
| Title: The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration |
| Format: eBook |
| Number of Pages: 221 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: February 2021 |
| File Size: 6mb |
| Read The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration |
 |

The Depression In Freezing Point Of 0 01 M Aqueous Ch 3 Cooh Solution Is 0 02046 1 M
| Title: The Depression In Freezing Point Of 0 01 M Aqueous Ch 3 Cooh Solution Is 0 02046 1 M |
| Format: PDF |
| Number of Pages: 271 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: May 2018 |
| File Size: 5mb |
| Read The Depression In Freezing Point Of 0 01 M Aqueous Ch 3 Cooh Solution Is 0 02046 1 M |
 |

The Depression Of Freezing Point For 1m Urea 1m Glucose 1m Sodium Sulphate And 1 M Sodium Chloride Brainly In
| Title: The Depression Of Freezing Point For 1m Urea 1m Glucose 1m Sodium Sulphate And 1 M Sodium Chloride Brainly In |
| Format: eBook |
| Number of Pages: 160 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: July 2017 |
| File Size: 5mb |
| Read The Depression Of Freezing Point For 1m Urea 1m Glucose 1m Sodium Sulphate And 1 M Sodium Chloride Brainly In |
 |

Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of
| Title: Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
| Format: ePub Book |
| Number of Pages: 137 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: July 2019 |
| File Size: 1.1mb |
| Read Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
 |

Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of
| Title: Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
| Format: PDF |
| Number of Pages: 284 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: January 2019 |
| File Size: 2.2mb |
| Read Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
 |

The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration
| Title: The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration |
| Format: PDF |
| Number of Pages: 141 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: September 2017 |
| File Size: 3mb |
| Read The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration |
 |

Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of
| Title: Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
| Format: eBook |
| Number of Pages: 273 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: December 2020 |
| File Size: 1.6mb |
| Read Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
 |

Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl2 Are In The Ratio Of 1 1 2 3 2 Brainly In
| Title: Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl2 Are In The Ratio Of 1 1 2 3 2 Brainly In |
| Format: ePub Book |
| Number of Pages: 138 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: February 2019 |
| File Size: 6mb |
| Read Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl2 Are In The Ratio Of 1 1 2 3 2 Brainly In |
 |

The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration
| Title: The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration |
| Format: eBook |
| Number of Pages: 161 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: September 2018 |
| File Size: 1.1mb |
| Read The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1m Nacl Are In The Ration |
 |

The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1 M Nacl Are In The Ratio
| Title: The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1 M Nacl Are In The Ratio |
| Format: PDF |
| Number of Pages: 136 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: May 2019 |
| File Size: 5mb |
| Read The Depressions In Freezing Point For 1 M Urea 1 M Glucose And 1 M Nacl Are In The Ratio |
 |
Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of
| Title: Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
| Format: ePub Book |
| Number of Pages: 261 pages Depression In Freezing Point For 1 M Urea |
| Publication Date: June 2017 |
| File Size: 5mb |
| Read Depression In Freezing Point For 1 M Urea 1 M Nacl And 1 M Cacl 2 Are In The Ratio Of |
 |
On the other hand KCl dissociates into K and Cl- ions so that a 1 M KCl solution would be approximately 20 M in solute particles and the freezing point should be about 38 deg-C less than that of pure water. Correct option is. Depression in freezing point is a colligative property which depends on the number of particles present in the solution.
Here is all you need to learn about depression in freezing point for 1 m urea Correct option is. The depression in freezing point for 1 m urea 1 m glucose and 1 m NaCl are in the ratio. Depression in freezing point for 1 m urea 1 m nacl and 1 m cacl are in the ratio of1 1. Depression in freezing point for 1 m urea 1 m nacl and 1 m cacl 2 are in the ratio of the depressions in freezing point for 1 m urea 1 m glucose and 1 m nacl are in the ratio the depressions in freezing point for 1 m urea 1 m glucose and 1m nacl are in the ration depression in freezing point for 1 m urea 1 m nacl and 1 m cacl 2 are in the ratio of a 0 1m urea solution shows less depression in freezing point than 0 1m mgcl2 solution why quora the depression in freezing point of 0 01 m aqueous ch 3 cooh solution is 0 02046 1 m Watch Video in App.